Molten Aluminum
Effects of Non-Metallic Inclusions in Molten Aluminum
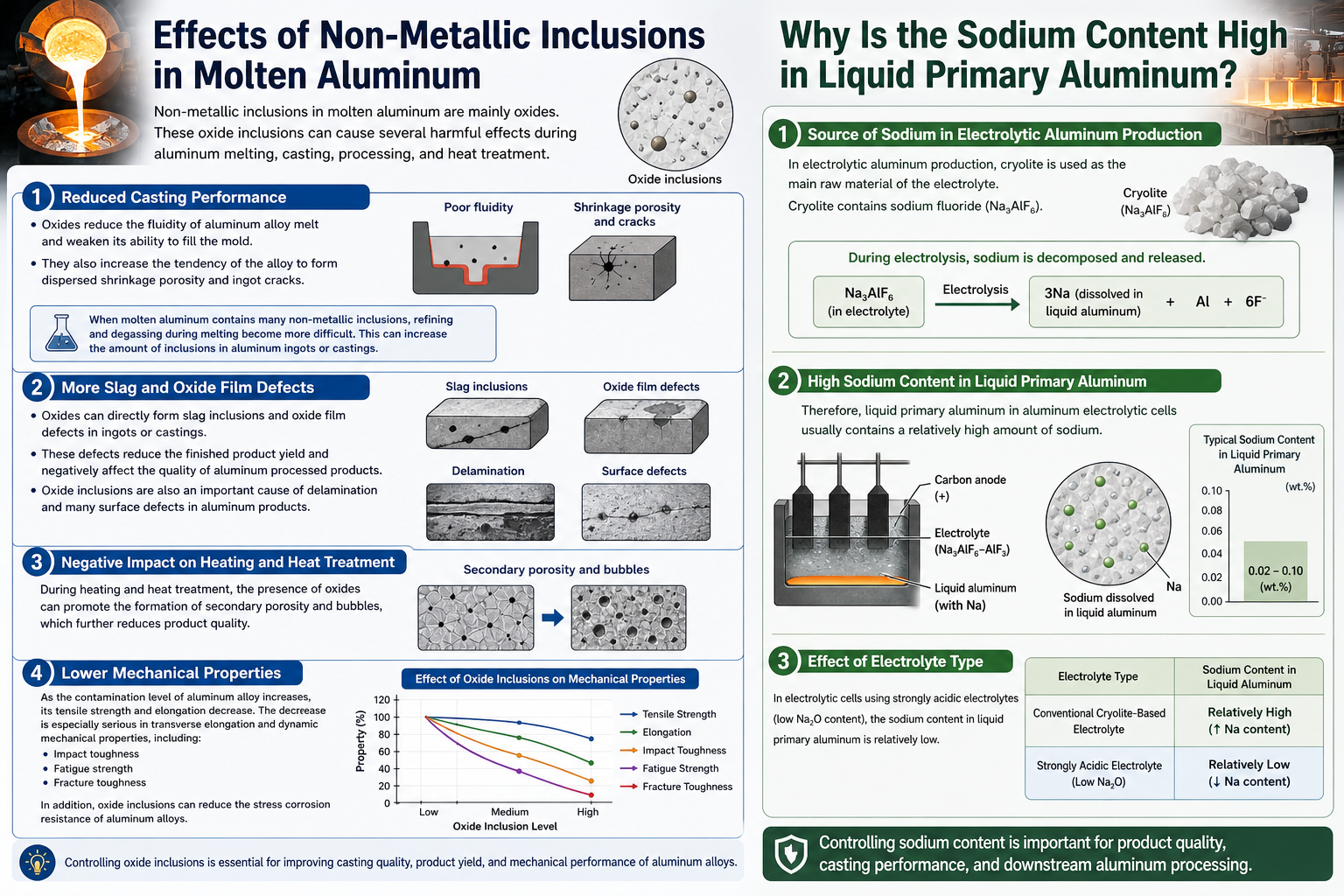
Non-metallic inclusions in molten aluminum are mainly oxides. These oxide inclusions can cause several harmful effects during aluminum melting, casting, processing, and heat treatment.
1. Reduced Casting Performance
Oxides reduce the fluidity of aluminum alloy melt and weaken its ability to fill the mold. They also increase the tendency of the alloy to form dispersed shrinkage porosity and ingot cracks.
When molten aluminum contains many non-metallic inclusions, refining and degassing during melting become more difficult. This can increase the amount of inclusions in aluminum ingots or castings.
2. More Slag and Oxide Film Defects
Oxides can directly form slag inclusions and oxide film defects in ingots or castings. These defects reduce the finished product yield and negatively affect the quality of aluminum processed products.
Oxide inclusions are also an important cause of delamination and many surface defects in aluminum products.
3. Negative Impact on Heating and Heat Treatment
During heating and heat treatment, the presence of oxides can promote the formation of secondary porosity and bubbles, which further reduces product quality.
4. Lower Mechanical Properties
As the contamination level of aluminum alloy increases, its tensile strength and elongation decrease. The decrease is especially serious in transverse elongation and dynamic mechanical properties, including:
- Impact toughness
- Fatigue strength
- Fracture toughness
In addition, oxide inclusions can reduce the stress corrosion resistance of aluminum alloys.
Why Is the Sodium Content High in Liquid Primary Aluminum?
In electrolytic aluminum production, cryolite is used as the main raw material of the electrolyte. Cryolite contains sodium fluoride. During electrolysis, sodium is decomposed and released.
Therefore, liquid primary aluminum in aluminum electrolytic cells usually contains a relatively high amount of sodium.
In electrolytic cells using strongly acidic electrolytes, the sodium content in liquid primary aluminum is relatively low.

